This article explores key thermodynamics concepts from AP Chemistry Unit 6, focusing on energy exchange in chemical and physical processes.
Core Thermodynamics Concepts
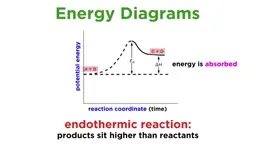
•Enthalpy (ΔH) measures energy change under constant pressure, with negative ΔH indicating exothermic reactions and positive ΔH indicating endothermic reactions.•Thermochemical equations include ΔH values that are stoichiometric, scaling with molar quantities and reversing sign for reverse reactions.•Heat transfer involves kinetic energy moving from hotter to colder objects until thermal equilibrium is reached.•Heat capacity depends on substance composition and amount, while specific heat (intensive property) is energy per gram per degree Celsius.Applications and Calculations
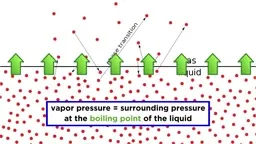
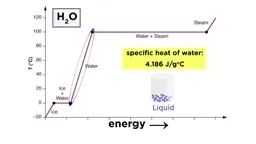
•Calorimetry uses devices like coffee cup calorimeters to measure heat exchange by combining temperature data with known heat capacities.•Phase changes have associated enthalpies: vaporization/condensation (ΔHvap), melting/freezing (ΔHfus), and sublimation/deposition, with heating/cooling curves showing temperature plateaus during transitions.•Bond enthalpies calculate ΔH by subtracting energy released from bonds formed from energy required to break bonds.•Enthalpy of formation (ΔHf°) measures energy to form one mole of compound from elements in standard states, used in ΔH = ΣΔHf°(products) - ΣΔHf°(reactants).Key Takeaways
•Enthalpy change (ΔH) quantifies heat exchange in reactions, with sign indicating exothermic (negative) or endothermic (positive) processes.•Specific heat and calorimetry enable precise measurement of heat transfer, while phase change enthalpies explain energy requirements for transitions like melting or vaporization.•Bond enthalpies and formation enthalpies provide methods to calculate reaction ΔH, essential for predicting energy changes in chemical systems.Conclusion
Mastering these thermodynamics principles allows for accurate analysis of energy flow in chemical reactions and physical processes.