This article summarizes key concepts from AP Chemistry Unit 9, covering thermodynamics and electrochemistry as explained in a concise review video.
Thermodynamics Fundamentals
•Entropy measures disorder: solids have lowest entropy, gases highest; temperature and volume increases raise entropy.•Gibbs Free Energy (ΔG) determines thermodynamic favorability: negative ΔG means favored, positive means unfavored; calculated via ΔG = ΔH - TΔS or from standard values.•Reactions with negative ΔH and positive ΔS are favored at all temperatures; endothermic reactions with decreasing entropy are unfavored at any temperature.•ΔG relates to equilibrium constant: negative ΔG yields large K, positive ΔG yields small K; kinetic control explains slow but favored reactions like rusting.Electrochemistry Applications
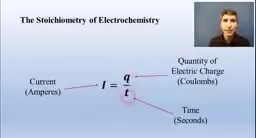
•Galvanic cells (batteries) harness redox reactions: anode is oxidation site, cathode is reduction site; electrons flow anode to cathode, cathode gains mass.•Cell potential (voltage) calculated from standard reduction potentials: E°cell = E°cathode - E°anode; positive voltage indicates favored process.•Nernst Equation adjusts for non-standard conditions: changing concentrations affects voltage; at equilibrium, voltage is zero (dead battery).•Electrolytic cells require external power for unfavored processes (e.g., metal plating); current, charge, and time relate via I = Q/t, with stoichiometry for mass calculations.Key Takeaways
•Gibbs Free Energy (ΔG) determines if a reaction is thermodynamically favored, with negative values indicating spontaneity and links to entropy and enthalpy changes.•Galvanic cells use redox reactions to generate electricity, with cell potential derived from standard reduction potentials and related to ΔG via equations.•Electrochemistry includes both spontaneous (galvanic) and non-spontaneous (electrolytic) processes, with calculations for voltage, charge, and mass based on stoichiometry.Conclusion
Mastering these thermodynamics and electrochemistry principles is crucial for success in AP Chemistry and understanding real-world chemical processes.