This article reviews the fundamental concepts of acids and bases, covering definitions, calculations, and practical applications like buffers and titrations.
Core Concepts and Definitions
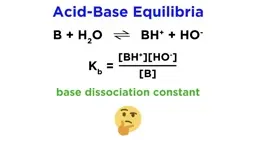
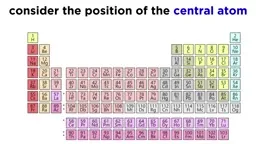
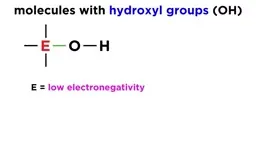
•Acids donate protons (H⁺) while bases accept them, as per the Brønsted-Lowry model, with conjugate pairs forming in reactions.•Strong acids and bases dissociate completely, whereas weak ones establish equilibria, described by Ka (acid dissociation constant) and Kb (base dissociation constant).•Acid strength depends on factors like conjugate base stability, periodic table trends (e.g., increasing acidity down and right), and molecular structure (e.g., electronegativity and oxidation state).Calculations and Applications
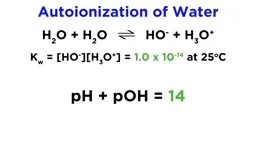
•pH and pOH quantify acidity/basicity: pH = -log[H₃O⁺], pOH = -log[OH⁻], with pH + pOH = 14 at room temperature.•Titrations involve stoichiometric neutralization, with curves differing for strong vs. weak acids; calculations use equilibrium concentrations to determine pH changes.•Buffers resist pH changes via weak acid-conjugate base pairs, analyzed using the Henderson-Hasselbalch equation: pH = pKa + log([A⁻]/[HA]).Key Takeaways
•Acid-base reactions involve proton transfer, with strength determined by conjugate stability and molecular factors like electronegativity.•pH and pOH calculations enable quantification of acidity, essential for understanding titrations and buffer solutions.•Buffers maintain pH stability through equilibria, with capacity depending on concentrations and described by the Henderson-Hasselbalch equation.Conclusion
Mastering these principles provides a foundation for analyzing chemical behavior in solutions and practical applications.