This article reviews the core concepts of chemical kinetics from AP Chemistry Unit 5, covering reaction rates, rate laws, and reaction mechanisms.
Reaction Rates and Rate Laws
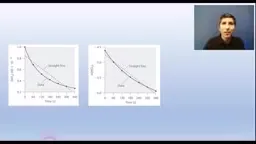
•Kinetics studies how fast reactions proceed, influenced by concentration, temperature, particle size, and catalysts.•Rate laws relate reactant concentrations to initial rates, with orders determined from experimental data (e.g., tripling concentration triples rate for first order).•Graphical methods determine order: concentration vs. time (zero order), ln(concentration) vs. time (first order), or 1/concentration vs. time (second order).•Integrated rate laws connect rate constants, time, and concentrations, with half-life equations specific to first-order processes.Reaction Mechanisms and Catalysis
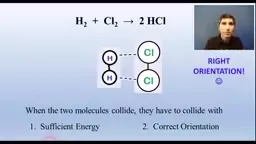
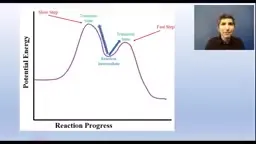
•Multistep reactions involve intermediates and catalysts, with the slowest step determining the overall rate law.•Activation energy and correct orientation are required for collisions, shown in energy profiles with peaks for transition states.•Catalysts lower activation energy and are not consumed, often participating in steps and regenerating, such as in surface catalysts.Key Takeaways
•Rate laws are derived from experimental data and describe how reactant concentrations affect reaction rates.•The slowest step in a multistep mechanism dictates the overall rate law and may involve intermediates.•Catalysts accelerate reactions by reducing activation energy without being consumed, often through participation in reaction steps.Conclusion
Mastering these kinetics principles is essential for understanding reaction dynamics in AP Chemistry.