This article reviews key concepts from AP Chemistry Unit 4 on chemical reactions, covering types of changes, equations, calculations, and reaction classifications.
Fundamentals of Chemical Reactions
•Physical changes involve alterations in appearance or state, such as phase changes or separating mixtures.•Chemical changes transform substances into new ones through bond breaking/forming, indicated by signs like gas production or color change.•Chemical equations must be balanced to conserve mass and atoms, with net ionic equations omitting spectator ions in solution reactions.Reaction Analysis and Calculations
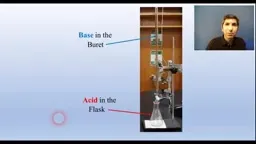
•Balanced equations serve as recipes for stoichiometric calculations using a three-step process: convert to moles, apply mole ratios from coefficients, and convert to final units.•Titrations, like acid-base experiments, determine equivalence points where moles of acid and base are equal, with indicators signaling endpoints.•Primary reaction types include acid-base (proton transfer), oxidation-reduction (electron transfer), and precipitation (solid formation from soluble ions).Advanced Concepts and Applications
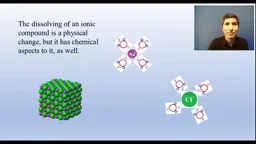
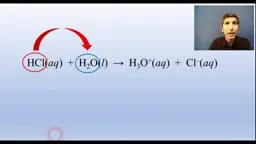
•The Brønsted-Lowry theory defines acids as proton donors and bases as proton acceptors, with conjugate pairs where stronger acids have weaker conjugate bases.•Redox reactions are analyzed using half-reactions to track electron transfer, balancing charges and electrons to derive overall equations.•Dissolving ionic compounds involves breaking ionic bonds and forming ion-dipole interactions with water, blurring lines between physical and chemical changes.Key Takeaways
•Chemical reactions involve bond changes and can be identified by signs like precipitate formation or temperature shifts.•Stoichiometry relies on balanced equations for calculations, with titrations and limiting reactants as common applications.•Reactions are categorized as acid-base, redox, or precipitation, each with specific mechanisms and analysis methods.Conclusion
Mastering these concepts prepares students for AP Chemistry exams by emphasizing reaction identification, equation balancing, and practical calculations.