Understanding Intermolecular Forces Through AP Chemistry Problems
Kaynak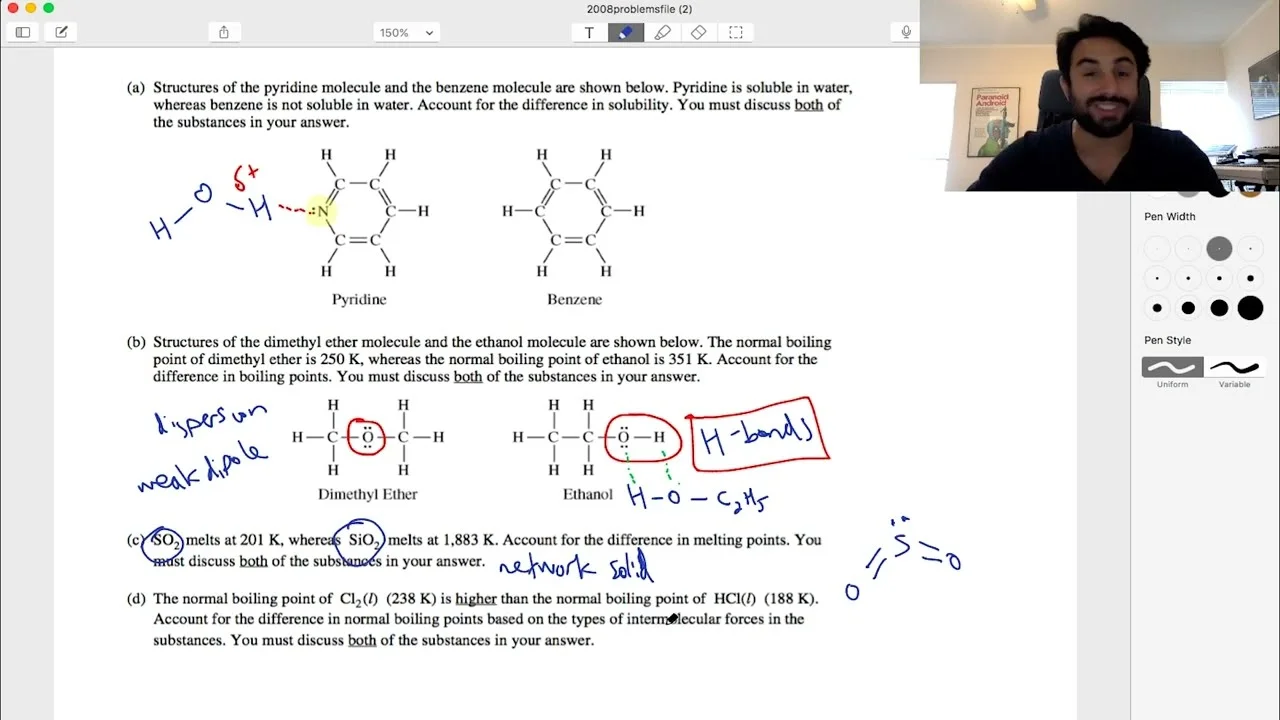
This article explores key concepts in intermolecular forces by analyzing four distinct chemistry problems. Each scenario demonstrates how different molecular structures and interactions affect physical properties like solubility and boiling points.
Solubility Differences: Purine vs. Benzene
The first problem examines why purine dissolves in water while benzene does not. This difference stems from molecular polarity and interaction capabilities. Purine contains a nitrogen atom with a lone pair, creating polar N-C bonds and enabling hydrogen bonding with water molecules. Water's polar nature allows it to form electrostatic interactions with purine through dipole attractions and hydrogen bonds. In contrast, benzene is a non-polar hydrocarbon lacking these polar features, preventing significant interaction with water. The principle 'like dissolves like' explains this solubility disparity, as only polar or hydrogen-bonding molecules can effectively engage with water's dipole.
Boiling Point Variation: Dimethyl Ether vs. Ethanol
Despite being structural isomers with identical molecular formulas (C2H6O), dimethyl ether and ethanol exhibit vastly different boiling points. Dimethyl ether boils at 250K, while ethanol boils at 351K. This discrepancy arises from intermolecular forces, not mass. Ethanol's hydroxyl group enables hydrogen bonding between molecules, creating strong electrostatic attractions that require substantial energy to overcome for vaporization. Dimethyl ether, as an ether, lacks direct O-H bonds and cannot form hydrogen bonds; it relies on weaker dispersion forces and slight dipole interactions. The stronger hydrogen bonds in ethanol lower its energy state in the liquid phase, necessitating more heat to liberate molecules into the gas phase.
Melting Points: SO2 vs. SiO2
Sulfur dioxide (SO2) melts at 201K, whereas silicon dioxide (SiO2) melts at a dramatically higher temperature. This contrast highlights different bonding types. SO2 consists of discrete molecules held together by intermolecular forces, specifically dipole-dipole interactions, which are relatively weak and easily disrupted. SiO2, however, is a network solid with a lattice of covalent silicon-oxygen bonds throughout. Melting SiO2 requires breaking these strong covalent bonds, not just overcoming intermolecular attractions. The energy needed to sever multiple covalent bonds in the extensive network far exceeds that for separating SO2 molecules, accounting for SiO2's much higher melting point.
Boiling Point Comparison: Cl2 vs. HCl
Chlorine (Cl2) has a higher normal boiling point than hydrogen chloride (HCl), which may seem counterintuitive given HCl's capacity for dipole-dipole interactions. While HCl can engage in these stronger electrostatic forces, Cl2's larger size and greater polarizability lead to more significant dispersion forces. The chlorine atom in Cl2 has a much larger electron cloud than hydrogen in HCl, resulting in stronger momentary and induced dipoles. Although dipole-dipole interactions are generally stronger than dispersion, the substantial increase in dispersion forces for Cl2 outweighs this advantage. Mass differences also contribute, but the key factor is the dominance of dispersion in Cl2 due to its size.
Analyzing Intermolecular Force Strengths
These examples underscore the importance of comparing relative strengths among intermolecular forces. Hydrogen bonding typically surpasses dipole-dipole interactions in strength, as seen in ethanol versus dimethyl ether. Covalent bonds in network solids are far stronger than any intermolecular force, explaining SiO2's high melting point. However, exceptions occur when dispersion forces become dominant due to large molecular size, as with Cl2 versus HCl. Recognizing which forces a substance can exhibit—dispersion, dipole-dipole, or hydrogen bonding—and assessing their magnitudes is crucial for predicting physical behaviors. This analysis requires both identification and comparative evaluation of interactions.
Key Takeaways
Conclusion
These chemistry problems demonstrate how intermolecular forces dictate physical properties through molecular structure and interaction types. Mastering these concepts enables accurate predictions of solubility, boiling points, and melting behavior in various substances.




